via Vanderbilt University
Researchers at Vanderbilt University Medical Center have relieved symptoms in a mouse model of Rett syndrome with a drug-like compound that works like the dimmer switch in an electrical circuit.
The finding, reported this week in the journal Human Molecular Genetics, suggests a potential new way to treat the rare neurodevelopmental disorder, which occurs predominantly in females, as well as more common forms of autism spectrum disorder.
Further study is needed before this approach can be tested in patients, but animal work from other groups suggests that symptoms in Rett syndrome are reversible, said senior author Colleen Niswender, Ph.D., associate professor of Pharmacology and director of Molecular Pharmacology in the Vanderbilt Center for Neuroscience Drug Discovery (VCNDD).
Rett syndrome patients exhibit a constellation of symptoms, including verbal and nonverbal communication deficits, difficulty walking, progressive developmental regression, epilepsy and intellectual disability. The hallmark symptom is repetitive hand movements such as wringing, washing, clapping or tapping.
Most cases of Rett syndrome occur spontaneously from random mutations in the MECP2 gene, which results in disruptions in neurotransmission, including signals mediated by the excitatory transmitter glutamate. A related genetic disorder, termed MECP2 Duplication syndrome, results from overexpression of the MeCP2 protein and is found predominantly in young boys.
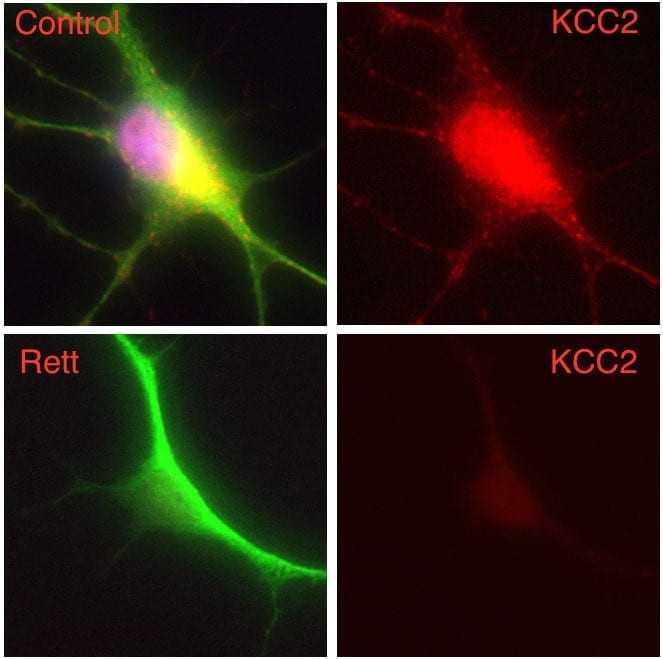
The researchers used a mouse model in which MECP2 was “knocked out,” resulting in Rett syndrome-like symptoms. Expression of the metabotropic glutamate receptor 5 (mGlu5), which is important in transmitting nerve signals in the brain, was reduced in brain areas of these mice.
The investigators also found reduced levels of mGlu5 expression in autopsy samples from the brains of Rett syndrome patients, suggesting that the animal work may translate to the clinical population.
They then tested a compound, developed at Vanderbilt, called a positive allosteric modulator or PAM that, like a dimmer switch, “turns up” the activity of mGlu5 when glutamate binds to it.
Animals treated with the compound showed improved motor performance and reduced Rett syndrome-like symptoms, including repetitive clasping of their hind claws.
Previous studies have suggested that enhancing excitatory neurotransmission may increase an already high risk of seizures in Rett patients, but in the current study, seizure occurrence did not increase, even after multiple weeks of chronic dosing with the PAM.
This may be because the compound acts on only a subset of mGlu5-mediated signaling pathways, leaving others unaffected.
“Our work demonstrates that potentiating the activity of mGlu5 can rescue several deficits commonly found in mouse models of Rett syndrome,” said the paper’s first author, Rocco Gogliotti, Ph.D., a postdoctoral fellow in the VCNDD.
“Importantly, by integrating recent advancements in mGlu5 pharmacology into the drug development process, we were able to test the efficacy of our novel compound in a mouse model without observing negative side effects, such as seizures, that have been historically associated with this target,” he said.
Disease progression – and mGlu5 expression – change through time in MeCP2-deficient mice. “A very careful dissection of the time course of the disease is required before modulators of excitatory transmission can be advanced to clinical studies,” Niswender said.
“It may be that early stages of Rett syndrome will need to be treated differently than late-stage disease,” she said.
Learn more: Study reveals possible ‘dimmer switch’ drug for Rett syndrome
The Latest on: Rett syndrome
[google_news title=”” keyword=”Rett syndrome” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
- Rett Syndrome Research Trust Partners with Vivalink to Shift Symptom Assessment Paradigms in Rett Syndromeon April 23, 2024 at 6:00 am
Vivalink, a leading provider of digital healthcare solutions, announces its collaboration with the Rett Syndrome Research Trust (RSRT) to advance research into Rett syndrome, a rare neurological ...
- Vivalink and RSRT collaborate to improve Rett syndrome research with wearable technologyon April 23, 2024 at 5:52 am
In a major partnership aimed at accelerating research into Rett syndrome, a rare neurological disorder predominantly affecting girls, digital healthcare solutions company Vivalink, has joined forces ...
- Neurogene Announces Upcoming Presentation of Safety Data from Phase 1/2 Trial of NGN-401 Gene Therapy for Rett Syndrome at ASGCT Meetingon April 23, 2024 at 12:56 am
Title: Preliminary Safety Results from the Ph1/2 study of NGN-401, a Novel Regulated Gene Therapy for Rett Syndrome Time: 12:00 p.m. ET The abstract can be accessed at this link.
- Acadia's Rett syndrome drug under priority review in Canadaon April 22, 2024 at 7:18 am
SAN DIEGO - Acadia Pharmaceuticals Inc. (NASDAQ:ACAD) has announced that its New Drug Submission for trofinetide, intended to treat Rett syndrome, has been accepted by Health Canada, which has also ...
- Acadia's Rett syndrome drug under priority review in Canadaon April 22, 2024 at 2:14 am
SAN DIEGO - Acadia Pharmaceuticals Inc. (NASDAQ:ACAD) has announced that its New Drug Submission for trofinetide, intended to treat Rett syndrome, has been accepted by Health Canada, which has ...
- Understanding, Recognizing, and Treating Rett Syndromeon April 21, 2024 at 5:00 pm
The clinical picture of Rett syndrome is unique in that no other condition comprises a period of rapid mental and physical deterioration, followed by apparent stabilization or even improvement.
- Neurotech reports positive top-line Phase 1/2 Rett Syndrome clinical trial resultson April 16, 2024 at 10:23 pm
Neurotech Int (ASX:NTI) reports Phase 1/2 Rett Syndrome clinical trial results with NTI164 being the first broad-spectrum cannabinoid drug.
- Rett Syndrome: Model of Neurodevelopmental Disorderson April 14, 2024 at 5:00 pm
Rett syndrome is a neurodevelopmental disorder that primarily affects girls, most of whom have mutations in the transcription regulatory gene MECP2. However, mutations in MECP2 also have been ...
- Rett syndrome: the genetic disorder almost exclusively targeting girlson April 3, 2024 at 5:00 pm
If you haven't heard of Rett syndrome, it's probably because it's very rare. Affecting an estimated one in 12,000 baby girls born each year (and only rarely seen in boys), this genetic disorder ...
- The Researcher Hunting for a Rett Syndrome Cureon April 2, 2024 at 4:59 pm
Qiu hopes to start clinical trials on human subjects as soon as this fall, provided the drug is proven safe in primates, as he plays catch-up in the race to find new treatments for children affected ...
via Google News and Bing News